
Mortality and Cancer Risks, Which Drugs to Avoid & Better Alternatives
New April 15, 2019 - FDA Hypnotics Policy Reduces Global Warming:
A Satirical Look at the Bright Side

Mortality and Cancer Risks, Which Drugs to Avoid & Better Alternatives
By Daniel F. Kripke, M.D.
New April 15, 2019 - FDA Hypnotics Policy Reduces Global Warming:
A Satirical Look at the Bright Side

Sleeping Pills Could Shorten Your Life
WARNING: Sleeping pills are hazardous to your health and could cause death from cancer, infections, overdoses, respiratory failure, other illnesses or accidents.
For over 40 years, as a doctor and medical researcher, I have worked to assess the risks of sleeping pills. I have learned that sleeping pills are associated with significantly increased mortality. Sleeping pills kill people.
This means that people who take sleeping pills die sooner than people who do not use sleeping pills. On average, those taking sleeping pills die several years early.
Chapter 1 describes how sleeping pills cause cancer, illnesses, and deaths. Chapter 2 describes how sleeping pills fail to improve sleep substantially, and on average make function worse the next day.
I first became interested when I saw the work of Dr. E. Cuyler Hammond at the American Cancer Society. In 1975, I went to visit The American Cancer Society, starting a collaboration which lasted for many years. American Cancer Society data from over one million people showed that use of sleeping pills was associated with more deaths within six years, but insomnia by itself was not associated with any death risk.
As of July, 2018, there were at least 42 published studies of the mortality risks of sleeping pills. Of the 42 studies which reported either greater or lesser mortality associated with sleeping pills, 40 studies showed that people taking sleeping pills died sooner. (The last two studies were a mix of evidence that sleeping pills caused deaths in some ways and other evidence they did not.) For more medical details about mortality and other risks of sleeping pills, follow the link to a comprehensive review [1], and be sure to check updates.
Here is an example of these studies. From electronic medical records,[2] we studied over 10,000 patients who took sleeping pills and over 20,000 matched patients who did not take sleeping pills. The patients
who took sleeping pills died 4.6 times as often during follow-ups averaging 2.5
years. Patients who took higher doses (averaging over 132 pills per year) died
5.3 times as often. Even those patients who took fewer than 18 pills per year
had 3.6 times the deaths of patients who took no hypnotics. Other newer and even larger studies have more recently reported similar results.
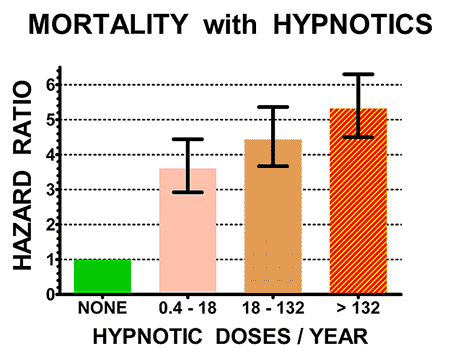
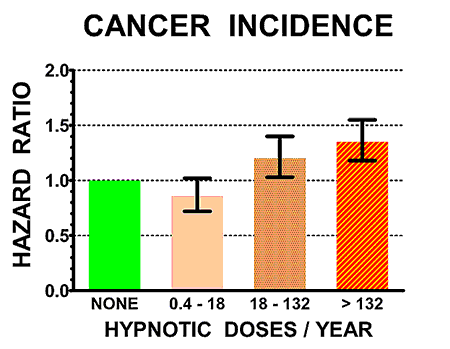
The illustrations above show the hazard ratios for mortality (above) and cancer incidence (below) for the control non-users of hypnotics (doses/year = NONE, in green) and for three groups of users of hypnotics with increasing numbers of doses/year prescribed. Hazard ratios above 1.0 are estimates of how many times the mortality rates or cancer incidence of sleeping-pill users exceeded that of controls. The heavy black bars show the statistical 95% confidence limits of the hazard estimates, that is, statistically the hazard ratio of the sample is 95% likely to be within the bars above and below the vertical black lines. However, unknown biases in the samples could produce true risks outside the confidence limits.
Patients who took sleeping pills died 4.6 times more often (on average) than patients who avoided sleeping pills.
Notice that people who took 18 pills a year or less (most no more than three pills) had substantially higher mortality. Our study and several more recent studies indicated that even one or two prescriptions might be lethal. The risk was especially high for people taking opioid pain pills, because sleeping pills increase the narcotic overdose risk. Sleeping pills are especially dangerous combined with narcotics, alcohol or both.
It seems quite likely that the sleeping pills were causing early death for many of the patients. In addition, those who averaged two to three sleeping pills per week or more were 35% more likely to develop a new cancer.
We made great efforts to match the patients taking sleeping pills with those not taking sleeping pills for age, sex, smoking history, and various measures of poor health, so it seemed to be a fair comparison. Nevertheless, it is true that finding that sleeping pill use is associated with early death does not by itself prove that the sleeping pills are causing those deaths. Theoretically, there could be confounding factors or biases in the selection of patients which caused those deaths without involving sleeping pills. We can only say that we found almost no evidence of such biases. Although there was certainly at least a small amount of confounding, it seemed to us unlikely that biases could entirely explain all of those excess deaths and cancers.
If sleeping pills cause even a small portion of the excess deaths and cancers associated with their use, they are too dangerous to use.
Some readers will remember when the cigarette companies claimed that the fact that cigarette smoking is associated with cancer and early death did not prove that cigarettes cause cancer. Cigarette manufacturers have by now given up on that argument. The risks are quite similar with sleeping pills. For absolute proof, we would need large randomized controlled trials of cigarettes or sleeping pills, but nobody is going to do such trials that now would be hard to make ethical. If the cigarette or sleeping pill companies believed that such trials would prove that their products were safe, they would have done such controlled trials many years ago.
The specific sleeping pills we studied were zolpidem (e.g., Ambien), temazepam (e.g., Restoril), eszopiclone (e.g., Lunesta), zaleplon (e.g., Sonata), other benzodiazepines such as triazolam (e.g., Halcion) and flurazepam (e.g., Dalmane), barbiturates, and sedative antihistamines such as diphenhydramine (e.g., Benadryl). Most of the patients in this study were taking zolpidem or temazepam. We had less data about the other drugs. However, all of the sleeping pills studied were significantly associated with excess mortality. Because of the way the study was done and its limited size, we could not say that one sleeping pill was safer than another.
Sleeping
Pills Associated with Significant Mortality Risk
Zolpidem
Temazepam
Eszopiclone
Zaleplon
Triazolam
Flurazepam
Estazolam
Quazepam
Barbiturates
(esp. phenobarbital)
Antihistamines, mainly diphenhydramine
These results do not necessarily apply to any sleeping pill which was not included in our study, except perhaps zopiclone (because zopiclone is half eszopiclone). Zopiclone is a sleeping pill popular outside the United States. Specifically, the risk data did not cover doxepin, ramelteon, melatonin, suvorexant, or trazodone, though some of those might also be unsafe.[3]
1.A. New sleeping pills cause cancer in animals
Were the epidemiologic studies just statistical accidents, or do sleeping pills really cause cancer? Several years ago, the Food and Drug Administration (FDA) started making available on the internet some of their documents about the review of those newer sleeping pills approved for marketing in the United States since 1998. You can find these documents yourself through the US Food & Drug Administration’s Online Service, Drugs@FDA.[4]
To my great surprise, I learned that rats and mice given high doses of zaleplon (Sonata), eszopiclone (Lunesta) as part of zopiclone, and ramelteon (Rozerem) developed cancer. The information available was a little vague to be certain, but it seems that these new sleeping pills all caused cancer in animals. FDA reviewers thought some of the results were worrisome. One of the reasons I am not sure I understand these results is that I cannot find that the companies have ever published the study details in the medical literature. It is conceivable that the manufacturers do not want these cancer experiments understood. Some of the drugs also broke chromosomes, which is a well-known specific chemical mechanism by which drugs cause cancer. The FDA has large animal testing facilities but has not seemed interested in checking if sleeping pills cause cancer.
There was also some older and confusing information about zolpidem (Ambien). Although one of the old records[5] seemed to say that animals given zolpidem developed three kinds of cancer, and FDA reviewers were concerned about the risks, the new labeling approved[6] for the extended release version of zolpidem (Ambien CR) says no evidence of carcinogenic potential was observed in either mice or rats. I would like to know how the company figured they do not owe people a warning. Some FDA scientists also wondered, according to internal documents I found.
1.B. Evidence that sleeping pills cause cancer in people
In 2005 and 2006, several new sleeping pills were introduced into the U.S. market. The industry was hoping to make several billion dollars a year. Because the companies wanted to market sleeping pills for long-term consumption, they did larger studies of long-term use than ever had been done before. Summaries of the data from these randomizing controlled trials can be found at the FDA internet site[7] for zaleplon (Sonata), eszopiclone (Lunesta), and ramelteon (Rozerem). It turned out that because zaleplon was compared to zolpidem as well as to placebo, there was a bit of zolpidem data available also.
I admit that it is hard to understand the details of these controlled trials from the data which FDA has made available, but fortunately, I persuaded the FDA to review their own files. According to the FDA, there were nine new skin cancers and four other cancers among study participants randomized to the sleeping pills, but zero new cancers among those who only received placebo. The best estimate would be that the cancer rate for participants randomized to sleeping pills was several times that of the luckier volunteers who received placebo. Because these data come from randomizing comparisons, they appear to be proof that new sleeping pills (as a group) cause cancer. However, the controlled trial data were not sufficient to prove that any specific sleeping pill or brand causes cancer.[8] Let’s put together the epidemiologic data, the animal data, and the data from combining these controlled trials for four drugs. The evidence is that a patient who takes any of the sleeping pills listed in the box above is increasing his or her risk of getting cancer. I feel that my patients should be warned about this risk.
We do not have clear evidence that one sleeping pill has more cancer risk than another. In our epidemiologic study, we only demonstrated statistically significant cancer risks specifically for zolpidem and temazepam, the most popular drugs in that study, but none of the other drugs for which we had less data were clearly any better or worse. For patients prescribed over 2-3 sleeping pills per week, there was a 35% increased risk of developing cancer within an average of 2.5 years.
A new study from Taiwan has appeared, based on a representative national health insurance data base.[9] These authors studied zolpidem, which was the most popular hypnotic in Taiwan and the United States. With over eight years of follow-up, the Taiwan authors found a considerably larger cancer hazard associated with zolpidem than our study found with shorter follow-up. There have been additional studies with similar results. [10]
1.C. More about lethal risks of sleeping pills
As a young medical student in my first year of training, one of the first things I learned in our student laboratory was that the humane way to “put an animal to sleep” was to administer a fatal dose of a barbiturate such as pentobarbital. A bit later, I learned that pentobarbital was being prescribed almost automatically as a sleeping pill for patients in the hospital. Pentobarbital and related drugs are currently used to execute the death penalty on prisoners. Any medical student knows that these drugs can kill.
Doctors have a wonderfully complete understanding of how sleeping pills such as pentobarbital kill animals. These drugs bind with protein molecules called GABA receptors on the surface of nerve cells. The same protein receptor molecules bind at the same time with a neurotransmitter chemical called GABA, which gives them their name. Barbiturates and other sleeping pills accentuate the action of GABA, which is to cause the receptor molecule to allow chloride ions to enter the nerve cells. Since the chloride ions are negatively charged, they make the inside the nerve cell more negatively polarized, which in turn, makes the nerve cells less likely to fire (to generate nerve activity). When the nerve cells which stimulate the muscles of breathing are overly inhibited from firing by sleeping pills, the animal stops breathing. When breathing stops, the animal dies within a few minutes from lack of oxygen. This same mechanism explains how sleeping pills kill people who take an overdose. Mixture with other drugs, particularly opioids, alcohol and other sedatives, greatly magnifies the risks, as do various medical conditions, possibly stopping breathing with a dose not intended to be lethal.
In the 1970’s, a new group of sleeping pills became popular, molecules which chemically are named benzodiazepines. The first sold as tranquilizers were chlordiazepoxide (Librium) and diazepam (Valium). Soon, the benzodiazepine flurazepam (Dalmane) was marketed as a sleeping pill, and flurazepam soon dominated the market. The main advantage of benzodiazepines is that they initially seemed less likely to produce acute overdose deaths than barbiturates.[11]
A third generation of new sleeping pills have been benzodiazepine agonists, which means that the chemical molecules may not have the benzodiazepine structure of drugs like Valium, but they act at the same brain receptors. Epidemiologic data have not confirmed that benzodiazepines are safer than barbiturates, perhaps because of how they are combined with other drugs and alcohol.
There is an age-old belief that sleeping pills might help depressed patients. Rather, sleeping pill manufacturers’ controlled trials proved that sleeping pills can cause depression.[12] In fact, the sleeping pills examined in one study seemed to double the rate of new depressions. Use of sleeping pills is very strongly associated with suicide from all causes.
Suicide, accidental overdose and cancer are probably not the most common ways in which sleeping pills kill, but the other ways are more poorly understood and less well documented. Here are some of the other possible mechanisms.
All approved sleeping pills can cause “hangover,” that is, they not only reduce the action potentials of our brain cells during sleep, but they can also reduce brain cell activity during the day.[13] This can make us sleepy, less alert, confused, and weak during the day. We will discuss psychological consequences of this hangover in the next chapter, but here the issue is impairments of survival. Falls are much more common among elderly people who are taking hypnotics.[14] Of patients given Lunesta, 10% had accidents as compared to 6% given placebo in one study, and falls were specifically more common with Lunesta.[15] Because several studies show that people who are responsible for automobile accidents are unusually likely to have sleeping pills in their blood[16], it is thought that hangover may often cause automobile accidents, as well as other fatal accidents. The publicity about Ambien zombies driving like sleep walkers provides some extremely vivid examples.[17]
In the last 20 years, physicians have become concerned about sleep apnea, a condition where there are pauses of breathing during sleep. Physicians suspect that sleep apnea can cause deaths during sleep. Not all studies agree, but several studies have found that when a person with sleep apnea takes sleeping pills, there are more pauses in breathing and the pauses last longer. I was surprised to learn in the FDA data how well-documented it is that zolpidem makes sleep apnea worse. Because sleeping pills risk making apnea worse, many experts recommend that people with apnea should not be given sleeping pills. The problem is that almost everybody above age 40 has some sleep apnea, and the majority of people over 65 would meet commonly-used criteria for a diagnosis of sleep apnea.[18] Therefore, a large proportion of people taking sleeping pills are making their apnea worse. Over a period of many years, anything which makes sleep apnea worse would be expected to cause high blood pressure, and therefore, to increase the risk of heart attacks, heart failure, and strokes.
A final concern regarding mortality is how people care for themselves. Because sleeping pills, like tranquilizers, reduce worry about possible threats and risks in our lives, it is possible that the hangover effects of sleeping pills would reduce people’s attentiveness in taking care of themselves.
Endnotes for Chapter 1
1. Kripke DF: Hypnotic drug risks of mortality, infection, depression, and cancer: but lack of benefit [version 3]. F1000Res. 2018, 5:918![]()
![]() . [return]
. [return]
2. Kripke DF, Langer RD, Kline LE. Hypnotics’ association with mortality
or cancer: a matched cohort study. BMJ Open. 2012;2:e000850![]()
![]() . [return]
. [return]
3. Kripke, DF Is suvorexant a better choice than alternative hypnotics? F1000Res. 4, 456. 2015.![]()
![]() . [return]
. [return]
4. Administered by the U.S. Food & Drug Administration, Center for Drug Evaluation and Research, Drugs@FDA “allows you to search for official information about FDA approved brand name and generic drugs and therapeutic
biological products.” www.accessdata.fda.gov/scripts/cder/drugsatfda/![]()
![]() . [return]
. [return]
5. US Food & Drug Administration new drug application (NDA) 19-908 (Ambien) memos and exclusivity summary, previously downloaded at FDA website.[return]
6. Sanofi-Aventis U.S. LLC, Prescribing Information, Ambien CR (zolpidem
tartrate extended-release) tablets - CIV, October 2010, Publication No. ACR-WFPLR-WPLR-OCT10, www.accessdata.fda.gov/drugsatfda_docs/label/2010/021774s010lbl.pdf ![]()
![]() . [return]
. [return]
7. Administered by the U.S. Food & Drug Administration, Center for Drug Evaluation and Research, Drugs@FDA “allows you to search for official information about FDA approved brand name and generic drugs and therapeutic
biological products.” www.accessdata.fda.gov/scripts/cder/drugsatfda/![]()
![]() . [return]
. [return]
8. Kripke, DF. Possibility that certain hypnotics might cause
cancer in skin. J. Sleep Res. (2008) 17![]() , 245-250. [return]
, 245-250. [return]
9. Kao, C. H., Sun, L. M., Liang, J. A., Chang, S. N., Sung, F. C., and Muo, C. H. Relationship of zolpidem and cancer risk: a Taiwanese population-based cohort study. Mayo Clinic Proceedings![]()
![]() 87(5), 430-436. 2012. [return]
87(5), 430-436. 2012. [return]
10. Kripke DF: Hypnotic drug risks of mortality, infection, depression, and cancer: but lack of benefit [version 3]. F1000Res. 2018, 5:918![]()
![]() . [return]
. [return]
11. Institute of Medicine. Sleeping Pills, Insomnia, and Medical Practice. National Academy of Sciences, Washington, D.C., 1979. [return]
12. Kripke, D. F., Greater incidence of depression with hypnotic use than with placebo, BMC Psychiatry 7:42.![]()
![]() 2007. [return]
2007. [return]
13. Woods, JH et al. Benzodiazepines: Use, abuse, and consequences. Pharmacological Reviews. 1992;44:151-347. PDF available at ResearchGate![]()
![]() . [return]
. [return]
14. Tinetti, ME et al. Risk factors for falls among elderly persons living in the community. N.Engl.J.Med. 1988;319(26):1701-1707. [return]
15. FDA Medical Review of Lunesta, page 2, Center for Drug Evaluation
and Research Approval Package for Application No. 21-476, available as a PDF document
at the FDA website, www.accessdata.fda.gov/drugsatfda_docs/nda/2004/021476_Lunesta_medr.PDF![]()
![]() . [return]
. [return]
16. Hemmelgarn, B et al. Benzodiazepine use and the risk of motor vehicle crash in the elderly. JAMA. 1997;278:27-31.; Betts, TA et al. Effect of two hypnotic drugs on actual driving performance next morning. Br.Med.J. 1982;25 Sept:285-852. [return]
17. Liddicoat, Laura J. and Harding, Patrick. Ambien®: Drives Like
a Dream? Case Studies of Zolpidem-Impaired Drivers in Wisconsin, presentation
to the 58th annual meeting of the American Academy of Forensic Sciences, Washington
State Convention and Trade Center, Seattle, Washington, February 23, 2006. Powerpoint
slides from the presentation are available at the New York Times website
at www.nytimes.com/packages/other/business/Ambien.2-23-061.ppt![]()
![]() . [return]
. [return]
18. Kripke, DF et al. Prevalence of sleep disordered breathing in ages 40-64 years: A population-based survey. Sleep. 1997;20:65-76.; Ancoli-Israel, S et al. Sleep disordered breathing in community-dwelling elderly. Sleep. 1991;14(6):486-495. [return]
Table of Contents
The Dark Side of Sleeping Pills, in all its formats, including this eBook, copyright ©1997-2019 by Daniel F. Kripke, M.D. All rights reserved.